 
Bladder inflammation also affected somatic sensitivity at nearby (hindpaw) but not distant (forepaw) sites. That is, bladder inflammation produced significant colon hypersensitivity of a magnitude not dissimilar to that produced by intracolonic treatments that produce colon hypersensitivity ( 19). Colon sensitivity was assessed quantitatively by measuring the visceromotor response to colorectal distension (15–60 mmHg), which in CYP-treated mice was significantly increased relative to saline-treated controls ( Fig. 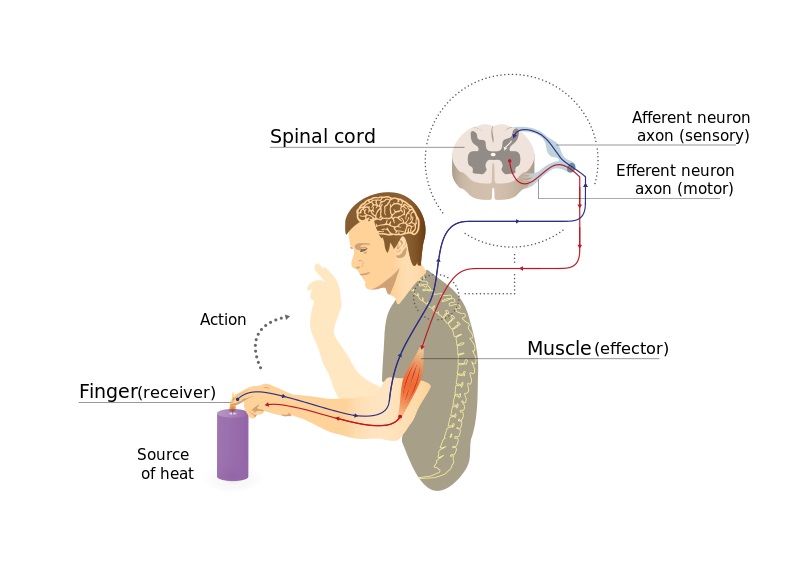
We next examined the effects of CYP-produced cystitis on sensitivity in an adjacent viscus, the colon. 
1 B), and increased the micturition frequency ( Fig. 1 A), lowered the peak micturition pressure ( Fig. CYP treatment shortened the latency to the first micturition ( Fig. In control (saline treated) mice, a saline infusion into the bladder led to filling with a slow pressure increase, eventually triggering a bladder contraction followed by visible micturition. We first established that the CYP treatment protocol employed produced bladder inflammation and altered bladder function. On the basis of these results, we examined the following four hypotheses: 1) visceral inflammation sensitizes visceral afferent pathways innervating different organs and converging onto the same spinal segments (viscerovisceral convergence) 2) visceral inflammation sensitizes somatic afferent pathways in areas of pain referral (viscerosomatic convergence) 3) somatic inflammation sensitizes visceral afferents converging onto the same spinal segments (somatovisceral convergence) and 4) transient overexpression of NGF in visceral or somatic structures sensitizes afferent pathways comparably with the effects seen with inflammation.Įffects of cystitis on micturition, somatic sensory thresholds, and colorectal sensation. The expression of NGF increases during visceral inflammation and contributes to the development of visceral hypersensitivity ( 6, 7, 54). While initially described as an important factor in the development of nociceptive neurons, NGF continues to play an important role in maintaining and modulating the properties of sensory neurons in adult life ( 29). ( 44) reported that acute colitis triggers bladder hyperactivity in rats, providing experimental evidence for cross-talk between different pelvic viscera.Ī previous report ( 17) has demonstrated that visceral inflammation triggers hyperalgesia referred to the hindpaw, which can be attenuated by immune neutralization of nerve growth factor (NGF). Conversely, many patients with interstitial cystitis also suffer from functional bowel disorders ( 2, 14). Patients with functional bowel disorders often also complain about pelvic pain or symptoms consistent with interstitial cystitis ( 61). Interestingly, visceral pain syndromes affecting different organs often coexist. Visceral pain has several unique characteristics, including referral to distant somatic sites, which is due to viscerosomatic convergence of afferent pathways at the level of the spinal cord and higher centers within the central nervous system. In conclusion, sensitization of sensory pathways by inflammation or NGF contributes to the development of hypersensitivity in neighboring organs and cutaneous referral sites and provides a potential mechanism underlying the coexistence of pain syndromes in patients with functional diseases. Hindpaw inflammation and injection of the NGF- but not GFP-encoding viral vector or front paw inflammation induced mechanical and thermal hyperalgesia in the affected hindpaw and increased responses to colorectal distension without altering the micturition reflex. Cystitis and NGF but not GFP overexpression in the bladder triggered bladder hyperactivity associated with mechanical and thermal hypersensitivity in cutaneous referral sites and enhanced responses to colorectal distension. Thresholds to mechanical and thermal stimulation of the hindpaw, visceromotor responses to colorectal distension, and cystometrograms were performed in appropriate controls and mice with experimentally induced cystitis, inflammation of the hindpaw or front paw, or injection of viral vectors encoding NGF or green fluorescent protein (GFP). We examined whether inflammation or transient overexpression of nerve growth factor (NGF) in one tissue triggers hypersensitivity in referral sites. 
Sensory neurons innervating different tissues converge onto second-order neurons in the spinal cord. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
